
Product safety and sterilization standards are in the forefront of every manufacturing company’s mind. Pharmaceutical companies must adhere to stringent sterilization and safety standards.
What if…
What if you could attach or embed a tag or label to every single asset you had that would track everything about the item from creation until you were no longer responsible for it?
What if that same tag or label could withstand sterilization and rugged conditions?
Imagine if…
Imagine tossing all the pens and clipboards.
Imagine reducing the number of times your assets where handled by humans, reducing the chance for contamination.
Imagine the success of your production and inventory tracking.
The Solution is Here: RFID Tags that Can Be Sterilized
Tego, the leading innovator and pioneer in the creation and deployment of intelligent assets, has created the world’s only sterilization-proof ultra-high frequency (UHF) Radio-Frequency Identification (RFID) chip.
Tego’s products can withstand Gamma sterilization, eBeam sterilization, Ethylene oxide sterilization, and Cold storage (-80C).
But it gets even better.
This cutting-edge chip also has a dynamic software application behind it. You can capture the full digital history of any asset including its entire life cycle, workflow, chain of custody, sterilization and more. This history begins prior to manufacturing, spans its duration and aftermath, and captures critical data throughout the environmental monitoring process.
With the label or tag permanently affixed to the asset, the asset contains a living memory of its own history throughout its workflow. Even better, the label or tag can withstand multiple cycles of sterilization.
How is the Data Accessed?
The data is accessible via smart phone app, portable device (tablet, etc), or can be read using an autonomous reader that is placed strategically in one or multiple locations. The digital data collected on any asset is virtually unlimited.
It can include:
- batch, workflow step or stage
- explicit processes for compliance
- expiration dates
- any other attributes that are relevant and critical to the workflow
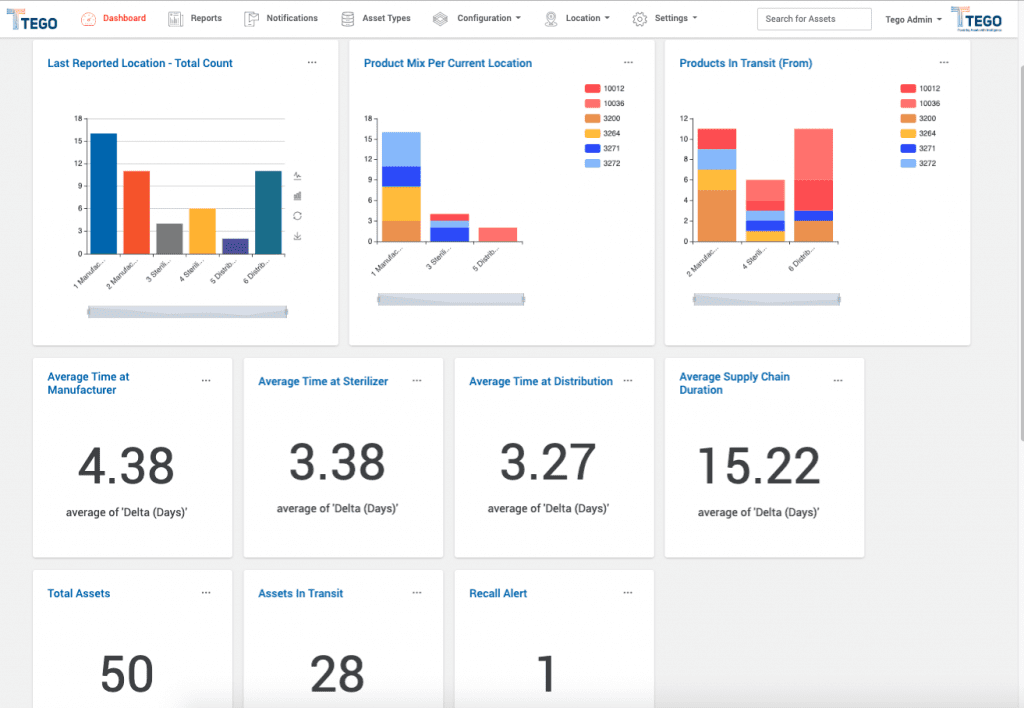
In addition to residing with the asset itself, the data is transmitted and backed up to the TegoHub, a centralized database and web-based application managed by the Tego platform. TegoHub can be configured to interface with other systems such as LIMS or BMS (building management), extending the reach and usefulness of the asset data.
Bottom Line: The Tego platform will integrate with what you are already using, saving time and money. 
More specifically, the platform can be configured to automate the following use cases:
- Digitization of manufacturing room logbooks
- Media life cycle for use during contamination events (used or unused media)
- Equipment usage
- Material/waste/product/equipment flows
- Chain of custody for batches and related documents
The solution enables automated tracking of materials, samples and assets for less labor-intensive activities, better visibility and reporting, and reduces overall contamination risk on the manufacturing floor.
Sound Interesting? Give PaladinID a Call to Find Out More!
Benefits of RFID Tags That can be Sterilized
QUALITY CONTROL
- Automate data collection from devices and people
- Eliminate redundancy data entry and transcription errors
- Gain direct traceability of QC Micro programs
- Improve worker efficiency
- Improve regulatory compliance
- Make sound product and quality release decisions
OPERATIONAL
- Accurate, secure, and automated digitized data flowing directly from the manufacturing room floor into LIMS, BMS, MES, etc
- Automated alerting and event reporting based on workflow movement of aseptic materials, samples, and assets
- Personnel monitoring in real time
ROI
- Digitizing materials, samples and assets
- Fifty percent reduction in labor
- Improved regulatory compliance
- Reduced risk
- Reduced financial penalties
- Better reporting for FDA
How Do I Find Out More?
PaladinID is excited to announce that we are among a very few select companies who are privileged to partner with and promote Tego’s cutting edge products and solutions. Please contact us if you would like to more about the ways Tego can help you become even better at asset tracking, controlling, and monitoring.
Without this solution manual paper and pencil processes are burden with:
- paper scheduling
- market labeling of sample media
- manual reconciliation
- paper log book entry
- manual notification of deviations
- test results stored in shelves of paper binders
All of these things are a challenge to navigate in an audit. Tego solves all of this for you.
Send us an Email or give us a call at 888.972.5234 for more information.
For more information on PaladinID
Get Help With Your Next Label Project
We have over 35 years of providing exceptional service and labeling products to the world. Take the first step to an easy, stress-free solution for your label needs by contacting us.
Schedule a call below or email dritchie@paladinid.com
Make Your Mark
“Making companies more competitive by offering the correct label printing solution, on time, within budget, while creating unmatched value”.
About PaladinID, LLC
PaladinID develops and supports high-performance barcode labeling applications. We work with our clients to “Make Your Mark” by providing the expertise and tools necessary to create an entire product label printing solution. Located in central New Hampshire, PaladinID has been serving Massachusetts, Vermont, Maine, Connecticut, Rhode Island, New England, and beyond for over 30 years, and in 2017, became an RFID-certified company. We look forward to working with you.
You may be interested in our other services:
Product Labels
Product Labels
Labels for every type of application: Blank, Pre-printed, Variable data
Label Printers
Label Printers
We sell and support: Direct/thermal transfer, Inkjet, Laser
Printer Ribbons
Printer Ribbons
We sell ribbons for ALL barcode printers including: Zebra, Datamax, Sato, Intermec
Flexible Packaging
Flexible Packaging
We offer a wide variety of packaging containers for your products.
Label Software
Label Software
Software for all barcode printing and product labeling.
Label applicators
Label Applicators
Wide selection of applicators: Desktop/Mobile, Applicator only, Print & apply

